5.1 Gas Laws
SAP-7.A Explain the relationship between the macroscopic properties of a sample of gas or mixture of gases using the ideal gas law.
5.1a Boyle's Law Verification Lab
5.1b Empirical Properties of Gases
5.1e Molar Volume of a Gas Lab
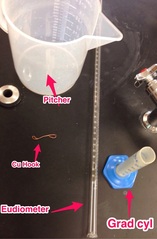
The goal is to determine the volume of one mole of hydrogen gas at STP. Hydrogen gas is generated by reacting magnesium ribbon with excess hydrochloric acid in a tube filled with water. (See materials and video below). The volume of hydrogen gas will be measured by water displacement after adjusting to atmospheric pressure and converting data obtained at lab conditions to STP conditions, utilizing...
|
5.1f Molar Mass of a Volatile Liquid
Determine, by experimentation, the molar mass of different volatile liquids utilizing vapor density measurements.
|
More Resources...
|
This is a playlist of 8 separate videos.
|
|
|
|