4.2a Lewis Structures & Formal Charge
|
|
|
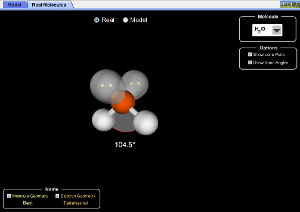
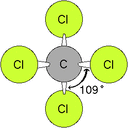
4.2b VSEPR & Molecular Geometry
from intro.chem.okstate.edu
Molecular shape is determined by the repulsion of shared and unshared electrons (as determined by Lewis structures). The notes, animation and simulation, using this lab sheet (vsepr_phet_lab.docx) make a couple of points: first, geometry is predictable and measurable; second, unshared electrons have a greater repulsive force than shared pairs; thirdly, all molecular geometries are spin-offs of five electronic geometries.
The PhET is a great way to review electron domains, molecular shapes and angles associated with each.
The PhET is a great way to review electron domains, molecular shapes and angles associated with each.
4.2c Hybridization
Molecular orbitals cannot be explained by existing atomic orbitals; there's an overlap and hybridization that must occur. The number of orbitals required for the hybridization depends upon the number of bonds needing to be made.
|
|
|