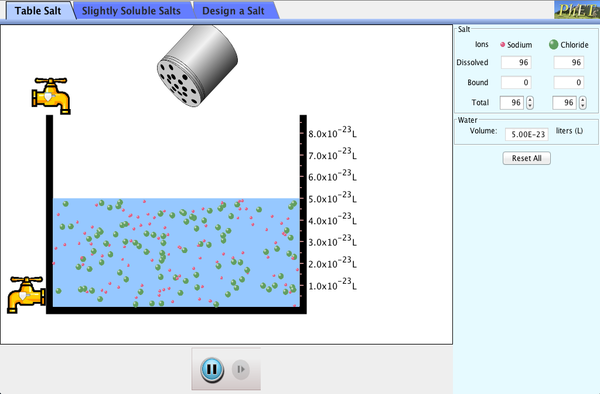
11.8 Salts & Ksp
LO 5.16 I can use Le
Chatelier's principle to make qualitative predictions for systems in
which coupled reactions that share a common intermediate drive formation
of a product.
LO 6.21 I can predict the solubility of a salt, or rank the solubility of salts, given the relevant Ksp values.
LO 6.22 I can interpret data regarding solubility of salts to determine, or rank, the relevant Ksp values.
LO 6.24 I can analyze the enthalpic and entropic changes associated with the dissolution of a salt, using particulate level interactions and representations.
LO 6.21 I can predict the solubility of a salt, or rank the solubility of salts, given the relevant Ksp values.
LO 6.22 I can interpret data regarding solubility of salts to determine, or rank, the relevant Ksp values.
LO 6.24 I can analyze the enthalpic and entropic changes associated with the dissolution of a salt, using particulate level interactions and representations.
|
(YouTube)
|
|
This is a review of solubility: Ksp is the equilibrium expression of insoluble/slightly soluble salts (precipitates).
Ksp is not solubility. |
Bozeman Science notes on solubility equilibria.
|
Notice in the latter part of the notes, there's a table with Kf (formation constant) for complex ions. As with all equilibrium constants, it can be compared with Q and you can predict the direction of the reaction.
Quiz #2: Sect.11.3