(Some Screencastomatic videos require Java 1.5 or later. Check your Java)
5.1 Bond, Bond, Bond, Bond (There's a joke here, I know it)
- Determine valence electrons based on electron configurations
- Explain why atoms form bonds
- Explain the role of periodic properties in predicting bond type and the resulting compound's possible properties
- Explain the role of electronegativity in determining bond type
- Classify bond type by electronegative difference
- Explain why most bonds are neither purely ionic or purely covalent
- Explain physical property differences in bond types, including melting points, conductivities, and solubilities
5.1a There's different Bonds?! (You know, Connery, Moore, Dalton, Brosnan and Craig)
Exploration Objectives
- What is the guiding question?
- How'd you do the lab? Why that way?
- What's your claim, evidence, and reasoning?
5.1b Ionic, Covalent & Metallic Bonds
|
(YouTube)
|
|
5.2 Lewis Structures/Diagrams
- Relate the octet rule to electron configurations and valence electrons
- Know the steps and be able to draw Lewis structures/diagrams for elements, ionic bonds, and molecules, including double and triple bonds
Quiz #2: 5.2
5.3 Molecular Geometry
- Explain VSEPR theory and the role of lone pairs vs. bonded pairs in molecular geometry
- Define the link between Lewis structures/diagrams and molecular geometry
- Predict shapes of molecules using VSEPR theory
- Relate electronegativity to molecule polarity
- Describe dipole-dipole and hydrogen bonding (i.e., intermolecular forces or IMFs), including their role in physical properties of molecules
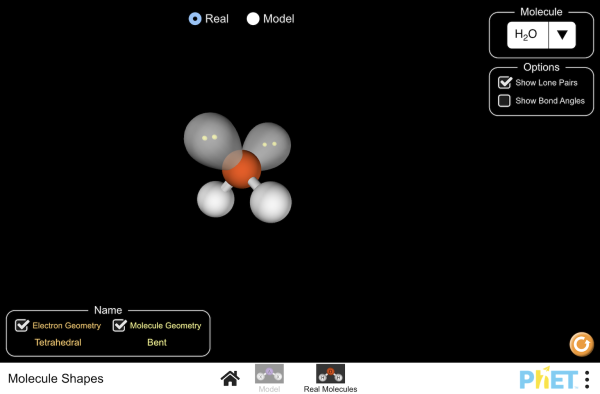
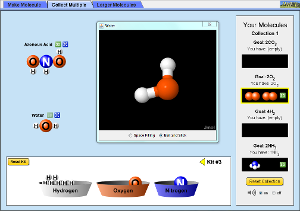
5.3a Explore Molecular Shape
Explore Objectives
- What question were you trying to answer and why?
- What did you do during your investigation and why did you conduct your investigation this way?
- What is your claim, evidence, and reasoning?
5.3b Build a Molecule PhET
5.3c VSEPR
5.4 Formulas & Nomenclature
- Write ionic and covalent chemical formulas and use proper nomenclature for each
5.4a Naming Ionic Compounds POGIL
5.4b Naming Molecular Compounds POGIL
5.4c Polayatomic Ions POGIL
5.4d Formulas & Nomenclature
Every chemical has a unique formula and a unique name. If you know one, you know the other. The left hand vodcast walks you through the basics of formulas and naming (some of which you may already know). The right vodcast adds basics about organic names/formulas and some practice going back and forth from formulas to names and names to formulas. You will need your flowcharts while you watch and work.
5.4d-1 Molecular Compounds
(YouTube)
|
|
5.4d-2 Ionic Compounds
(YouTube)
|
|
Technology Goals
- Students demonstrate creative thinking, construct knowledge, and develop innovative products and processes using technology. Students create original works as a means of personal or group expression. Students use of models and simulations to explore complex systems and issues.
- Students apply digital tools to gather, evaluate, and use information. Students locate, organize, analyze, evaluate, synthesize, and ethically use information from a variety of sources and media. Students process data and report results.
- Students demonstrate a sound understanding of technology concepts, systems, and operations. Students troubleshoot systems and applications and transfer current knowledge to learning of new technologies.
- Students use critical thinking skills to plan and conduct research, manage projects, solve problems, and make informed decisions using appropriate digital tools and resources. Students collect and analyze data to identify solutions and/or make informed decisions. Students use multiple processes and diverse perspectives to explore alternative solutions.
- Students understand human, cultural, and societal issues related to technology and practice legal and ethical behavior. Students exhibit a positive attitude toward using technology that supports collaboration, learning, and productivity. Students demonstrate personal responsibility for lifelong learning.